Leaching in metallurgy
Leaching in metallurgy refers to a process of extracting a desired metal from its ore or concentrate by using a liquid solvent. It is a common method employed in hydrometallurgical processes to recover valuable metals such as copper, gold, silver, uranium, and others.
The leaching process involves several steps:
Preparation: The ore or concentrate is usually crushed and ground to increase the surface area and expose the metal particles to the leaching solution.
Leaching: The crushed ore or concentrate is then mixed with a suitable liquid solvent, known as the leaching agent or leach solution. The leaching agent can be an acid, base, or other chemicals depending on the nature of the ore and the metal being extracted. The mixture is typically agitated or stirred to enhance the contact between the leaching agent and the metal particles.
Solvent-Metal Interaction: The leaching agent interacts with the metal, dissolving it into the solution. This process occurs through various chemical reactions, such as oxidation, reduction, or complexation, depending on the specific metal and leaching conditions.
Separation: After a sufficient leaching period, the leach solution, which contains the dissolved metal, is separated from the solid residue, known as the leach residue or “tailings.” Separation methods may include filtration, settling, or other solid-liquid separation techniques.
Recovery: The next step involves recovering the desired metal from the leach solution. This is typically achieved through additional processes, such as precipitation, solvent extraction, or electro-winning, depending on the specific metal and the desired purity level.
Leaching Types:
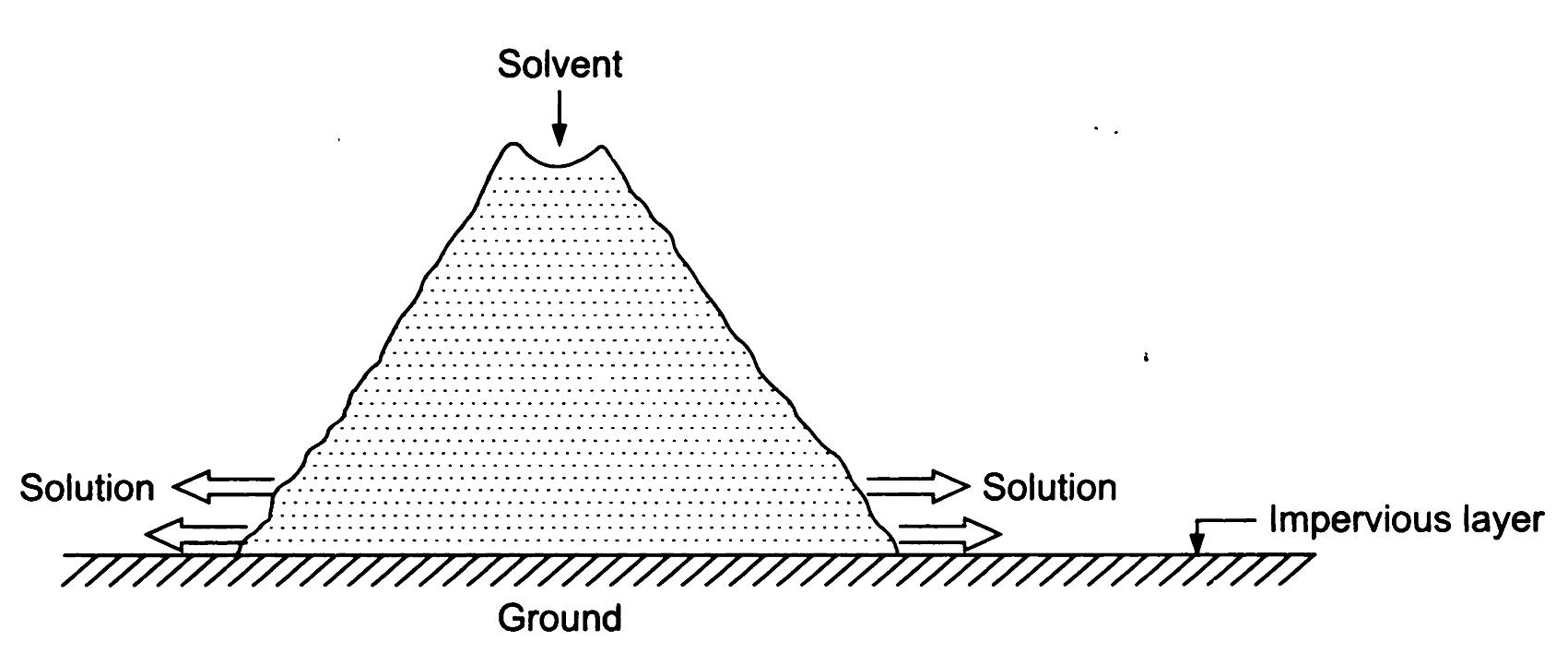
Heap Leaching: Heap leaching is a widely used method in which large piles or heaps of crushed ore are irrigated with the leaching solution. The solution percolates through the heap, leaching out the desired metal, and the pregnant solution is collected and processed for metal recovery. Heap leaching is often employed for low-grade ores or materials with large particle sizes.
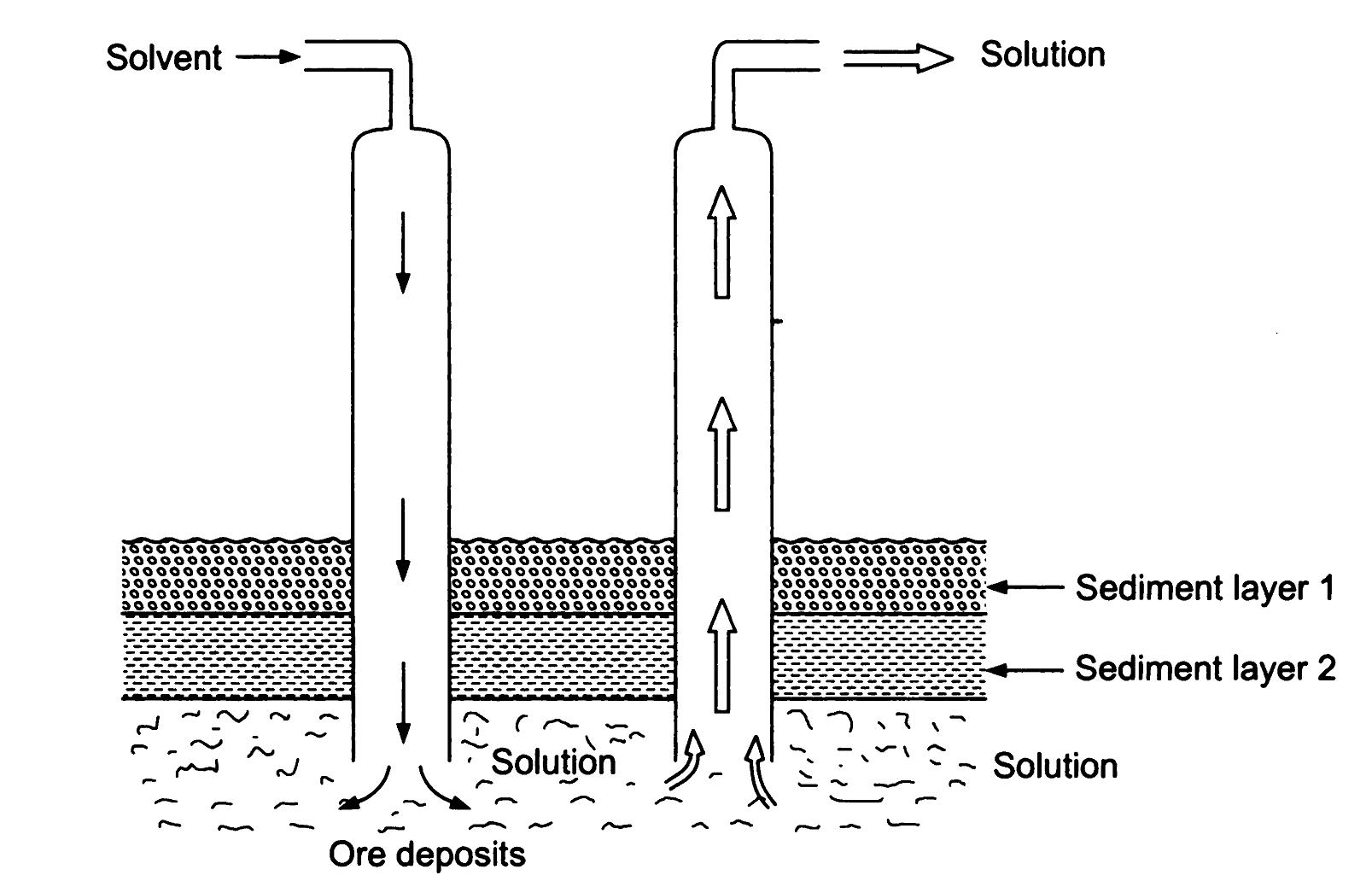
In Situ Leaching: In situ leaching, also known as solution mining or in-place leaching, is a technique used for recovering metals from underground ore bodies. Instead of extracting the ore and bringing it to the surface, the leaching solution is injected directly into the ore deposit. The solution dissolves the metal, and the pregnant solution is brought to the surface for further processing.
Environmental Considerations: Leaching processes, particularly those involving acidic solutions, can raise environmental concerns due to the potential for acid mine drainage. Proper management of the leach solutions and containment of the leach residues are crucial to prevent contamination of soil and water sources. Various environmental safeguards and monitoring systems are employed to minimize the impact of leaching operations.
Leaching of Other Materials: While leaching is commonly associated with metal extraction, it is also utilized in other fields. For example, in the agricultural industry, leaching is used to remove unwanted salts or minerals from soils to improve their productivity. In the pharmaceutical industry, leaching is employed for the extraction of active compounds from plant materials or the purification of pharmaceutical products.
Research and Development: Continuous research and development efforts are dedicated to improving leaching processes, optimizing leaching conditions, and developing more efficient leaching agents. This ongoing exploration aims to enhance metal recovery rates, reduce the environmental footprint, and expand the applicability of leaching to previously untapped resources.
Leaching is often preferred when the metal content in the ore is low or when traditional extraction methods, such as smelting or roasting, are not viable due to the nature of the ore or environmental considerations. It offers advantages such as lower energy requirements, reduced emissions of harmful gases, and the ability to process low-grade ores economically.
Overall, leaching plays a crucial role in metallurgy, enabling the extraction of valuable metals from ores and concentrates for various industrial applications.